Microbiome Vaginal: What It Is, Functions, Balance, and Tests
The vaginal microbiome is a topic of growing interest in medicine and…
Continue reading
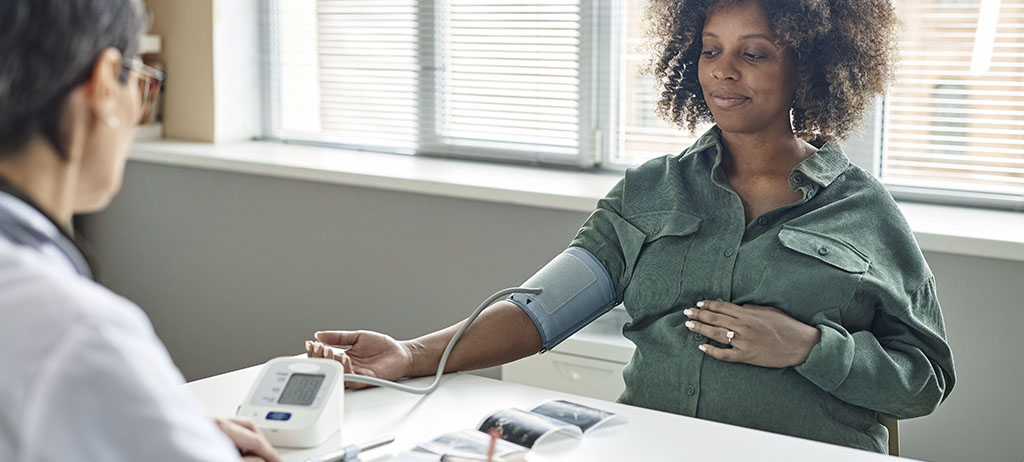

Preeclampsia is one of the most significant hypertensive disorders of pregnancy, with a substantial impact on maternal and neonatal health. It is estimated to affect 2% to 8% of pregnancies worldwide and is responsible for approximately 70,000 maternal deaths and 500,000 fetal and neonatal deaths annually, according to the World Health Organization (WHO).
Despite advances in prenatal care and obstetric management, early diagnosis and proper handling of the condition remain challenging—especially in settings with limited access to specialized care.
In this article, you’ll gain a deeper understanding of what preeclampsia is, its causes, symptoms, and the potential complications for both mothers and babies. We also explore how validated biomarkers and algorithms are transforming the diagnosis and management of this condition, enabling early and effective interventions. Keep reading to learn how preeclampsia can be prevented, diagnosed, and treated.
Preeclampsia is a multisystem hypertensive syndrome specific to pregnancy and is one of the leading causes of maternal and fetal morbidity and mortality (1, 2). It is characterized by elevated blood pressure (≥ 140/90 mmHg) and proteinuria (≥ 300 mg/24h) occurring after the 20th week of gestation (3).
In some cases, it may present without proteinuria and be identified through other signs of organ dysfunction, such as thrombocytopenia, elevated liver enzymes, renal insufficiency, pulmonary edema, or neurological manifestations like persistent headaches and visual disturbances.
Its pathophysiology involves endothelial dysfunction, an exaggerated inflammatory response, and abnormal placentation, resulting in inadequate placental perfusion and an increased risk of maternal-fetal complications (4).
According to the Preeclampsia Foundation, around 10 million women are affected by the condition annually, with approximately 76,000 maternal deaths related to preeclampsia and other hypertensive disorders of pregnancy, making it one of the leading causes of maternal and perinatal mortality worldwide (5).
Progression of the condition can lead to severe complications such as eclampsia (seizures associated with preeclampsia), HELLP syndrome (hemolysis, elevated liver enzymes, and low platelets), and fetal growth restriction.
Risk factors include a family history of preeclampsia, first pregnancy, obesity, diabetes, autoimmune disorders, and multiple gestation (6).
Eclampsia is a severe complication of preeclampsia, characterized by seizures occurring during pregnancy or in the postpartum period. While both conditions are linked to gestational hypertension, the presence of seizures is the key differentiating factor between eclampsia and preeclampsia.
It is a potentially fatal condition that requires immediate medical intervention. Without proper treatment, seizures may progress to coma, permanent neurological damage, or even maternal or fetal death (7).
Historically, preeclampsia was named as such because it was believed to precede eclampsia. Today, it is understood that seizures are just one of the potential manifestations of advanced disease.
Although usually a complication of severe preeclampsia, eclamptic seizures may occur even in the absence of clear clinical signs of the condition (7).
Preeclampsia is considered a placental-origin disorder with a two-stage development. The first stage involves abnormal placentation early in pregnancy, followed in the later trimesters by a “maternal syndrome” characterized by an excess of circulating antiangiogenic factors—substances released by the placenta that enter the maternal bloodstream, triggering inflammation, elevated blood pressure, and other typical symptoms of the condition (8, 9).
The exact reasons why placental development is abnormal in some pregnancies remain unclear. Animal model research suggests that inadequate oxygenation of the placenta (uteroplacental ischemia) may trigger the hypertensive response and multi-organ dysfunction typical of the clinical phase of the disease (10).
Several factors have been associated with placental dysfunction, including oxidative stress, alterations in natural killer (NK) cells at the maternal-fetal interface, and genetic and environmental influences. However, none have been conclusively proven in humans.
What is better understood is that an abnormal placenta releases soluble toxic substances into the maternal circulation, which induce systemic inflammation, endothelial dysfunction, and the hallmark clinical signs of preeclampsia (8, 9, 11).
Additionally, according to the International Society for the Study of Hypertension in Pregnancy, the condition is defined by a systolic blood pressure of ≥ 140/90 mmHg in (12):
Preeclampsia typically develops after the 20th week of pregnancy and may be asymptomatic or present with the following signs (13):
Eclampsia is the severe progression of preeclampsia and includes seizures. Additional symptoms may include:
Both conditions require urgent medical care, as they can endanger the lives of both mother and baby.
Preeclampsia can result in severe complications for both mother and baby. Without proper management, the condition can escalate to life-threatening situations (3).
Maternal complications include:
Preeclampsia can also pose risks to the baby, such as:
Strict maternal monitoring and early intervention are essential to reduce the risks of preeclampsia and prevent adverse outcomes for both mother and baby.
Risk assessment for the development of preeclampsia is traditionally based on the maternal medical history, considering factors such as nulliparity, high body mass index, gestational diabetes, history of preeclampsia, and chronic hypertension.
However, this clinical approach alone is able to identify only about 30% of women who will actually develop the disease (12). To improve this detection rate, studies have been investigating the role of maternal serum markers—such as free beta-hCG, pregnancy-associated plasma protein A (PAPP-A), placental protein 13 (PP13), inhibin A, placental growth factor (PlGF), and A disintegrin and metalloproteinase 12 (ADAM12)—as auxiliary tools in the prediction of preeclampsia as early as the first trimester (14).
Early identification of high-risk pregnancies allows for more effective prenatal interventions, optimizing management and potentially reducing the severity of the disease. In this context, the multicenter prospective ASPRE study evaluated 26,941 singleton pregnancies using a combined first-trimester screening.
Maternal factors, mean arterial pressure, uterine artery pulsatility index (UtA-PI), and serum levels of PAPP-A and PlGF were considered between 11 and 13 weeks of gestation. This approach was able to detect 76.6% of early-onset preeclampsia cases and 38.3% of total cases (15).
The American College of Obstetricians and Gynecologists (ACOG) classifies preeclampsia into mild and severe forms, based on the degree of hypertension, the presence of proteinuria, and systemic involvement.
For screening pregnant women at increased risk, ACOG recommends the exclusive use of clinical data, such as medical history and body mass index (BMI). Although simple, this approach presents a modest detection rate and a high false-positive rate, close to 70% (16).
In turn, the United Kingdom’s National Health Service (NHS) adopted similar criteria but with more stringent thresholds (17). This reduced the false-positive rate to approximately 10%, but unfortunately with a significant drop-in detection rate—estimated at only 40%—which is considered clinically unacceptable.
Faced with these limitations, the Fetal Medicine Foundation (FMF) in London proposed a more comprehensive algorithm that combines clinical information (history and BMI) with uterine artery Doppler (UtA-PI), mean maternal arterial pressure, and biochemical markers, especially placental growth factor (PlGF) (18).
This integrated approach represents a significant advance. While conventional screening methods detect only about 30% of preeclampsia cases, new predictive tests based on multiple markers have the potential to reduce the incidence by up to 89% among at-risk women.
Treatment for preeclampsia varies depending on severity and gestational age and may include the use of antihypertensive medications, magnesium sulfate to prevent seizures, and, in more severe cases, early delivery as the only definitive intervention (6).
Monitoring and management of preeclampsia involve controlling acute hypertension, preventing seizures, and using clinical strategies to aim for a full-term pregnancy. In patients diagnosed with preeclampsia near term (≥37 weeks), when the fetus is mature, delivery is an effective way to treat the condition and optimize pregnancy outcomes.
In preterm pregnancies, the decision to continue the pregnancy must be weighed against the risk of developing multisystem disorders—organ dysfunction caused by worsening preeclampsia that may result in complications such as eclampsia and HELLP syndrome (hemolysis, elevated liver enzymes, and low platelet count).
Preventing preeclampsia and eclampsia involves strategies aimed at early identification of risk factors, blood pressure control, and adopting practices that support maternal and placental health. Some preventive approaches include:
These measures are essential to reduce the incidence of preeclampsia and minimize complications. If the condition does develop, early intervention and proper treatment are critical for ensuring maternal and fetal health.
Using an algorithm that incorporates medical history and biochemical data, SYNLAB has developed two tests aimed at early identification and diagnosis of preeclampsia.
The Pre-eclampsia 1st Trimester Test estimates the risk of developing preeclampsia between the 11th and 13th weeks of gestation. It combines the analysis of PlGF (placental growth factor) levels with PAPP-A (pregnancy-associated plasma protein A), blood pressure measurements, and ultrasound parameters.
The Pre-eclampsia 2nd and 3rd Trimester Test differentiates between healthy pregnancies and those with preeclampsia. It combines the analysis of the sFlt-1/PlGF ratio (soluble fms-like tyrosine kinase-1 / placental growth factor), blood pressure, and proteinuria measurement. It is indicated from the second half of pregnancy onwards.
These tests help determine the risk of developing preeclampsia or allow for early diagnosis, supporting timely management and interventions to control maternal and fetal complications.
The Pre-eclampsia 1st Trimester Test is a screening tool to estimate the risk of developing preeclampsia between 11 and 13 weeks of pregnancy. Risk is calculated using a proprietary algorithm—the only one compatible and validated by the Fetal Medicine Foundation (FMF) guidelines—that integrates clinical information, ultrasound data, and serum biomarkers, including PlGF and PAPP-A, as well as mean arterial pressure.
Who should take this test?
Pregnant women between 11 and 13.6 weeks of gestation, especially those with one or more of the following risk factors:
The Pre-eclampsia 2nd and 3rd Trimester Test evaluates the risk of developing preeclampsia in later stages of pregnancy. Risk calculation is based on an algorithm that integrates PlGF levels, maternal blood pressure, and proteinuria analysis.
Testing is recommended from the 20th week of pregnancy onward.
Who should take this test?
Performing accurate and up-to-date tests is essential for precise diagnoses and the best treatment strategies. SYNLAB is here to help.
We offer high-quality diagnostic solutions with strict quality control for companies, patients, and doctors. Present in Brazil for over 10 years, we operate in 36 countries across three continents and are the leading provider of diagnostic services in Europe.
Contact the SYNLAB team and learn more about the available tests!
References
1) Sibai B, Dekker G, Kupferminc M. Pre-eclampsia. Lancet. 2005;365(9461):785–799.
2) Steegers EA, von Dadelszen P, Duvekot JJ, Pijnenborg R. Pre-eclampsia. Lancet. 2010;376(9741):631–644.
3) Rana S, Lemoine E, Granger JP, Karumanchi SA. Preeclampsia: Pathophysiology, Challenges, and Perspectives. Circ Res. 2019 Mar 29;124(7):1094-1112.
4) Magee, L. A., Brown, M. A., Hall, D. R., Giguère, Y., Hébert, J., & Rey, E. (2022). The hypertensive disorders of pregnancy. Best Practice & Research Clinical Obstetrics & Gynaecology, 82, 32-50.
5) Preeclampsia Foundation. Disponível em: https://www.preeclampsia.org/
6) American College of Obstetricians and Gynecologists (ACOG). (2020). Hypertension in pregnancy. Obstetrics & Gynecology, 135(6), e237-e260.
7) Preeclampsia Foundation. Disponível em: https://www.preeclampsia.org/what-is-eclampsia
8) Redman CW, Sargent IL. Latest advances in understanding preeclampsia. Science. 2005;308:1592–1594. doi: 10.1126/science.1111726.
9) Romero R, Chaiworapongsa T. Preeclampsia: a link between trophoblast dysregulation and an antiangiogenic state. J Clin Invest. 2013;123:2775–2777. doi: 10.1172/JCI70431.
10) Palei AC, Spradley FT, Warrington JP, George EM, Granger JP. Pathophysiology of hypertension in pre-eclampsia: a lesson in integrative physiology. Acta Physiol (Oxf). 2013;208:224–233.
11) Roberts JM, Taylor RN, Musci TJ, Rodgers GM, Hubel CA, McLaughlin MK. Preeclampsia: an endothelial cell disorder. Am J Obstet Gynecol. 1989;161:1200–1204.
12) Brown MA, Lindheimer MD, de Swiet D, Van Assche A, Moutquin JM. The classification and diagnosis of the hypertensive disorders of pregnancy: statement from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Hypertens Pregnancy. 2001;20(1):IX-XIV. PMID: 12044323 DOI: 10.1081/PRG-100104165
13) Ramos JGL, Sass N, Costa SHM. Preeclampsia. Rev Bras Ginecol Obstet. 2017 Sep;39(9):496-512.
14) Audibert F, Boucoiran I, An N, Aleksandrov N, Delvin E, Bujold E, Rey E. Screening for preeclampsia using first-trimester serum markers and uterine artery Doppler in nulliparous women. Am J Obstet Gynecol. 2010 Oct;203(4):383.e1-8.
15) Rolnik DL, Wright D, Poon LCY, Syngelaki A, O’Gorman N, de Paco Matallana C, Akolekar R, et al. ASPRE trial: performance of screening for preterm pre-eclampsia. Ultrasound Obstet Gynecol. 2017 Oct;50(4):492-495.
16) Wilbert S Aronow. Hypertensive disorders in pregnancy. Ann Transl Med. 2017;Jun;5(12):266.
17) National Health Service (NHS). Disponível em: https://www.england.nhs.uk/
18) Fetal Medicine Foundation. Disponível em: https://fetalmedicine.org/
19) World Health Organization (WHO). (2021). Calcium supplementation during pregnancy for the prevention of pre-eclampsia and its complications.
The vaginal microbiome is a topic of growing interest in medicine and…
Continue reading
The human papillomavirus (HPV) is considered the leading cause of…
Continue reading
Female hormones play a central role in virtually every stage…
Continue reading