BRCA+16 GENES
Prevention of hereditary
gynecological cancer
Why undergoing this examination?
Hereditary gynecological cancer is characterized by its occurrence in individuals under 40 years of age. In women, it accounts for 5-15% of cases of this type of cancer. The origin of this genetic susceptibility often lies in mutations in the BRCA1 and BRCA2 genes, although there are other genes related to breast, ovarian, and endometrial cancer that should be studied to provide more comprehensive information for a broader medical analysis.
What is this exam?
The BRCA+ test is a patient’s DNA analysis to detect point mutations, small insertions/deletions, and copy number variations (CNVs) in 18 genes. These genes include ATM, BRCA1, BRCA2, BRIP1, CDH1, CHEK2, EPCAM, MLH1, MSH2, MSH6, NBN, PALB2, PMS2, PTEN, RAD51C, RAD51D, STK11, and TP53, all related to hereditary gynecological cancer. Additionally, in case of a positive CNV result for the BRCA1, BRCA2, and EPCAM genes, confirmation is done through MLPA.
For whom is it indicated?
- Women over 30 without prior history, as a preventive measure for breast and/or ovarian cancer;
- Women with a family history of breast (male or female) and/or ovarian cancer;
- Family members carrying a mutation in BRCA1 or BRCA2;
- Patients with this type of tumor, to determine their possible hereditary profile.
Technology
Next-generation sequencing (NGS).
BRCA+16 GENES
Prevention of hereditary
gynecological cancer
Hereditary gynecological cancer is characterized by its occurrence in individuals under 40 years of age. In women, it accounts for 5-15% of cases of this type of cancer. The origin of this genetic susceptibility often lies in mutations in the BRCA1 and BRCA2 genes, although there are other genes related to breast, ovarian, and endometrial cancer that should be studied to provide more comprehensive information for a broader medical analysis.
The BRCA+ test is a patient’s DNA analysis to detect point mutations, small insertions/deletions, and copy number variations (CNVs) in 18 genes. These genes include ATM, BRCA1, BRCA2, BRIP1, CDH1, CHEK2, EPCAM, MLH1, MSH2, MSH6, NBN, PALB2, PMS2, PTEN, RAD51C, RAD51D, STK11, and TP53, all related to hereditary gynecological cancer. Additionally, in case of a positive CNV result for the BRCA1, BRCA2, and EPCAM genes, confirmation is done through MLPA.
- Women over 30 without prior history, as a preventive measure for breast and/or ovarian cancer;
- Women with a family history of breast (male or female) and/or ovarian cancer;
- Family members carrying a mutation in BRCA1 or BRCA2;
- Patients with this type of tumor, to determine their possible hereditary profile.
Next-generation sequencing (NGS).
Advantages
SYNLAB GROUP
Guaranteed by the experience of the absolute European leader in laboratory diagnostics.
COMPREHENSIVE
Complete genetic study of genes related to hereditary gynecological cancer.
COMPLETE
- Complete genetic study of genes related to hereditary gynecological cancer;
- Genetic counseling in cases where it is necessary to assess the implications of the result for the patient and their family;
- Any pathogenic or uncertain significance mutation is confirmed through a new DNA extraction followed by Sanger sequencing, ensuring 100% coverage of all DNA bases read.
Extra Information
DOCUMENTATION – Available on the SYNLAB Direct for clients
- Informed Consent;
- Clinical Questionnaire;
- Medical Request.
PREPARATION
- Fasting is not necessary for the test.
Additional Information
Check below the list of genes analyzed in BRCA+16 GENES:
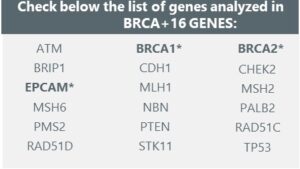
*Confirmation by MLPA in case of CNV detection.

Delivery Time
15 business days

Sample Type
8mL of total blood in EDTA
Or
Oragene OG510 Saliva